Because Fabry is an X-linked disorder and women have two X chromosomes, the GLA gene that causes Fabry may work normally in some parts of the body but not others. This is believed to be one reason why women with Fabry have more variable symptoms than men.
Women with Fabry may experience all or just some of the effects of Fabry to different extents, or they might never feel any of them. Because symptoms can vary, it is important that you and your healthcare provider recognize that your symptoms can be caused by Fabry and address them appropriately.

See the common ways that Fabry can affect women.
If you are a woman with Fabry disease who is experiencing symptoms, you are in the majority.

of women diagnosed with Fabry report having signs and symptoms of the disease.
Though women may first notice effects of Fabry when they are teenagers, they often have to wait years before finding out the cause.

is the average amount of time between when women first notice effects of Fabry and when they receive a definitive Fabry diagnosis through laboratory testing.
As mentioned above, women can experience the effects of Fabry just as severely as men. For example, one survey looking at pain intensity, duration, and frequency as a result of Fabry disease in both genders showed that pain ratings from women were virtually indistinguishable from those of men.
However, regardless of if or how you are experiencing its symptoms, Fabry is a progressive disease, meaning damage is always happening even if you can’t see it. That’s why it’s so important to keep an eye on your disease, as it can inform what steps you and your healthcare team take in managing your Fabry disease.
Hear from Rebecca, a Galafold patient and mother to a son with Fabry, who challenged a reluctance to diagnose and manage her Fabry because of her gender.
Hear from Rebecca, a Galafold patient and mother to a son with Fabry, who challenged a reluctance to diagnose and manage her Fabry because of her gender.
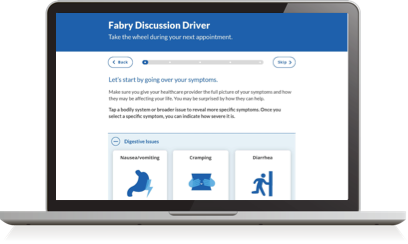
Take the wheel at your next appointment and cover what matters most to you.
Start nowFinding the right treatment for you starts with speaking up.
*Required fields
Sign up to get more information about Galafold and Fabry disease.
Before taking Galafold® (migalastat), tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take medicines or supplements containing caffeine as these medicines or supplements may affect how Galafold works.
How should I take Galafold?
Do not eat food, or take or drink any product that contains caffeine at least 2 hours before and 2 hours after taking Galafold to give a minimum 4 hour fast.
What are the possible side effects of Galafold?
The most common side effects of Galafold include headache, stuffy or runny nose and sore throat, urinary tract infection, nausea, and fever.
These are not all the possible side effects of Galafold. Call your healthcare provider for medical advice about side effects.
What is Galafold?
Galafold is a prescription medicine used to treat adults with Fabry disease who have a certain genetic change (variant) in the galactosidase alpha gene (GLA) that is responsive (amenable) to Galafold.
It is not known if Galafold is safe and effective in children.
You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Amicus Therapeutics at 1-877-4AMICUS.
Please click here for Full Prescribing Information, including Patient Information and Instructions for Use.
